You have no items in your shopping cart.
Search results for: 'CCL2'
- Featured
 Human CCL2 Protein, hFc Tag [orb1291052]Featured
Human CCL2 Protein, hFc Tag [orb1291052]Featured
Unconjugated
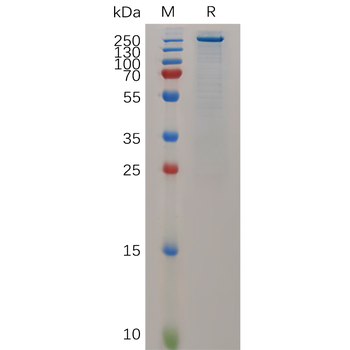
The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining.
The protein has a predicted molecular mass of 34.8 kDa after removal of the signal peptide. The apparent molecular mass of CCL2-hFc is approximately 35-55 kDa due to glycosylation.
Mammalian
100 μg, 10 μg, 50 μg - Recombinant Human CCL2 Protein [orb3002375]
SDS-PAGE: Greater than 95% as determined by reducing SDS-PAGE.
Predicted: 8.7 KDa. Observed: 13 KDa, reducing conditions
10 μg, 50 μg, 1 mg, 500 μg - Recombinant Mouse CCL2 Protein [orb3002391]
SDS-PAGE: Greater than 95% as determined by reducing SDS-PAGE. (QC verified)
Predicted: 8.5 KDa. Observed: 12 KDa, reducing conditions
500 μg, 1 mg, 10 μg, 50 μg - Featured
 Featured
Featured
>90% as determined by SDS-PAGE
15/18 kDa
20 μg, 100 μg, 500 μg - Recombinant Mouse CCL2/MCP-1 Protein, C-His [orb2964129]
ELISA, SDS-PAGE, WB
>90% as determined by SDS-PAGE.
16.75 kDa
1 mg, 50 μg, 100 μg